Infusion pump for single use
key word:
Classification:
Anesthesia products
- Product Description
-
Product name: Infusion pump for single use
Product model: CBI, CBI+PCA
Model
CBI
CBI+PCA
Filling volume
100mL, 150mL, 200mL, 275mL
Nominal flow rate
2mL/h, 3mL/L, 4mL/h, 5mL/L
Automatic liquid supply volume
/
0.5mL/times
Automatic liquid feeding and refilling time
/
15min
Product scope of application: This product is suitable for continuous injection of liquid medicine in clinical micro administration treatment.
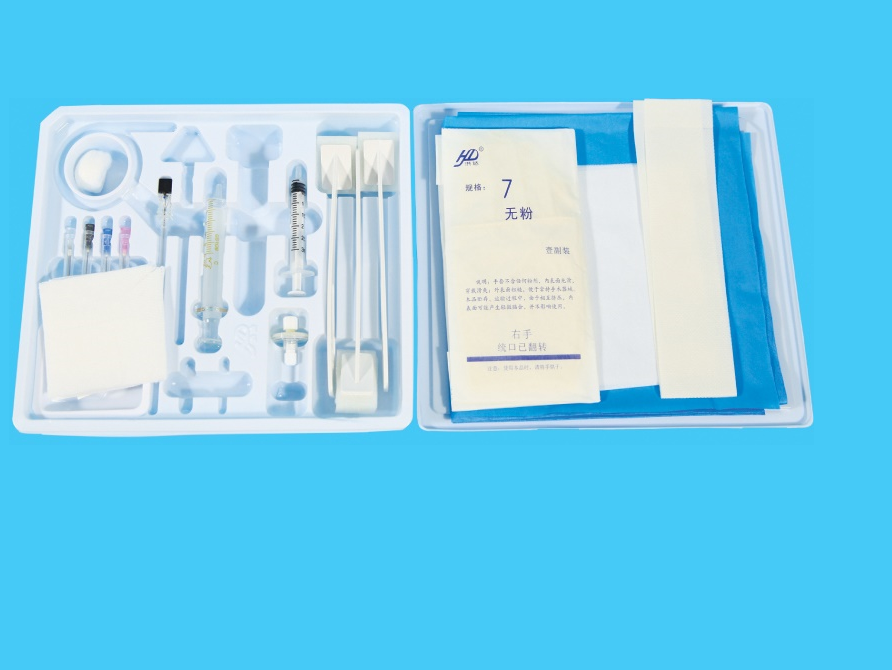
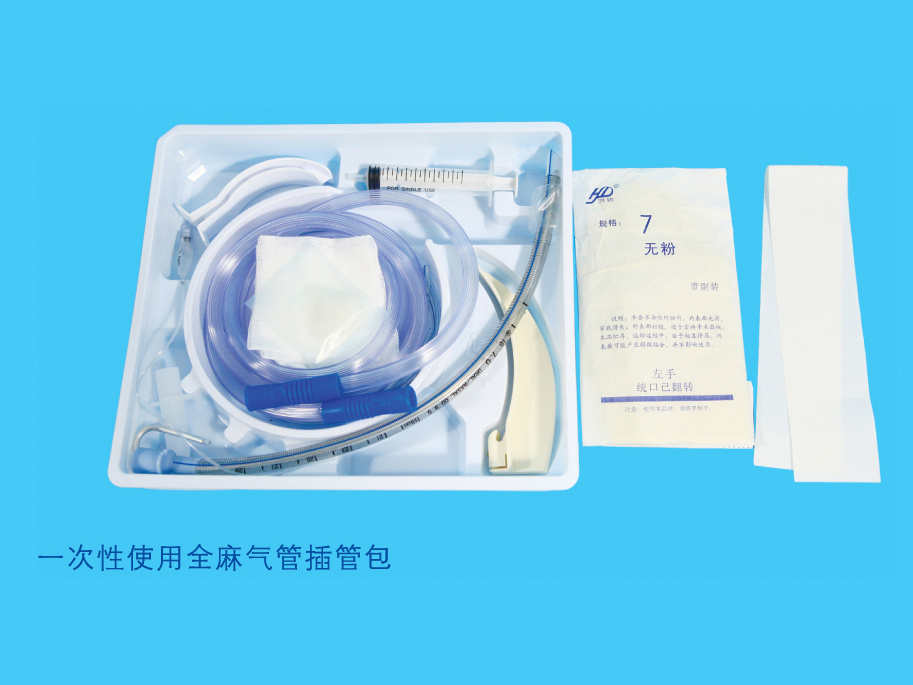
Product structure composition: It is consists of dosing port three-way, protective cap, protective bottle, liquid reservoir, and liquid medicine filter (5μm), flow limiting device, automatic control liquid feeding device (PCA), tubing, flow stop clamp and outer cone connector. The product is sterilized by ethylene oxide.
Indications:
For pain relief after surgery, such as:
− General surgery: Such as gallbladder removal, appendectomy, gastrointestinal surgery, etc.
− Joint replacement surgery: Such as analgesic management after knee or hip surgery.
− After fracture repair: Such as analgesic treatment after spinal surgery.
− After thoracic surgery: Such as analgesia after heart surgery and lung surgery.
− After obstetric surgery: Caesarean section.
Contraindications:
−Coma or disturbance of consciousness.
−Cognitive dysfunction, inability to correctly understand and use PCA technology.Precautions:
a.The single package is damaged and the protective sleeve falls off, it is prohibited to use
b.It is prohibited to use it beyond the validity period;
c.This product is disposable, please destroy it after use;
d.When using this product, please inject liquid medicine according to the filling capacity, and it is prohibited to inject too much liquid medicine;
e.Automatic control liquid feeding device (PCA) shall not be pressed without restraint, otherwise excessive dosage will cause harm;
f.During the first 1-2 hours of product use, the flow rate will be slightly faster than the nominal flow rate (still within the scope of this product standard), which is determined by the physical properties of the reservoir.
g.Due to the influence of such factors as drug concentration, working temperature and the position of the fluid reservoir, the actual flow rate and the nominal flow rate will change appropriately. The average flow rate deviation is within ±15% of the nominal flow rate, and the instantaneous flow rate deviation is within ±50% of the nominal flow rate;
h.Parallel infusion, flow rate variation due to temperature variation, flow rate variation due to partial fill or overfill of the reservoir.
i.During the application of PCA, adverse reactions should be monitored and dealt with in a timely manner;
j.The route of administration, the type and dose of the drug to be infused should be judged according to the specific conditions of the patient and clinical practice.
k.Before applying PCA analgesia, it is necessary to communicate with patients and their families. According to the different cultural levels of patients and their families, introduce in detail the advantages of PCA pumps and the importance of safe use. Train patients and families on how to use the equipment correctly, and emphasize that patients and families are prohibited from adjusting various parameters of the PCA pump by themselves, especially emphasizing that only the patient himself or the authorized person is allowed to press the PCA rescue
l.This product must be used by professional medical staff in medical institutions.
Obtain product quotation




 0791-85628888
0791-85628888 

 Email
Email 